Full-length human antibodies without fragment reformatting
Drug-like antibodies with reduced liabilities from the start
Real-time insight supports fast, informed decisions
85+ molecules advanced to clinic and 6 commercial therapies approved
At the core of Adimab’s yeast-based antibody discovery platform is a proprietary strain with more than 20 stable genetic modifications. These support high-quality expression of multi-domain biomolecules, including full-length IgGs and HCAbs, while maintaining stable diversity.
Featuring human IgG and HCAb libraries, plus complex biomolecules such as soluble TCRs, the strain provides a robust foundation for custom antibody discovery.
We leverage our proprietary synthetic IgG and HCAb libraries to provide state-of-the-art antibody diversity. IgGs are fully human, while HCAbs combine synthetic human frameworks with camelid-derived diversity. Incorporating AI/ML-driven in silico workflows in their design to maximize functional range, these non-overlapping libraries increase the probability of identifying potent, developable leads across all antibody discovery programs.
Our engineered yeast supports dual modes that streamline antibody discovery. Presentation mode enables enrichment of antibodies of interest using MACS and FACS, while secretion mode generates purified protein for analytical testing, functional validation, and lead identification.
Expression in yeast correlates with high yields in CHO and HEK cells, supporting efficient translation into fast, scalable antibody development.
Adimab’s antibody discovery platform accommodates a wide range of targets: soluble, membrane-associated, and membrane-spanning proteins, peptides, and even glycans. Our rigorous antigen QC process provides a robust foundation for successful discovery.
By combining antigen flexibility with yeast-based antibody discovery, we have discovered and engineered antibodies against hundreds of diverse targets over our rich history.

We direct discovery toward your desired profile with iterative MACS and FACS enrichment. Each round includes multiparameter assessment, which can include expression, affinity, cross-reactivity, pH dependence, and polyspecificity, so every decision is data-driven.
For difficult targets, such as membrane-obligate proteins, we apply novel immunization strategies in mice or llamas, followed by screening for single-cell or bulk immune diversity in our yeast platform.

With more than a thousand optimized antibodies, Adimab combines AI/ML-guided diversities, engineered yeast libraries, and real-time flow cytometry feedback to enhance specificity, potency, and developability.
Beyond IgGs, the platform supports engineering of external antibodies, fragments, multispecifics, and non-antibody proteins. This versatility ensures candidates are tuned for therapeutic performance across a range of molecular formats.

From each selection, hundreds of unique antibodies are expressed in yeast and purified for downstream analysis. Analytical assays span affinity determination across target forms, early developability metrics, epitope binning, and benchmarking against known antibodies.
Partners receive both protein and comprehensive data packages, enabling functional testing and informed lead identification with a strong experimental foundation.

Adimab is recognized for embedding developability early, with our foundational expertise advancing partner pipelines for over a decade. Rigorous evaluation of biophysical properties identifies candidates with favorable stability, scalability, and safety profiles before development.
This leadership is reflected not only in clinical molecules seeded from our platform but also in numerous peer-reviewed publications that set standards in the field.

Combining in vivo immunization with yeast presentation enables robust antibody discovery against membrane targets

Synthetic HCAb library enables rapid discovery of stable antibodies for multispecific T cell engagers
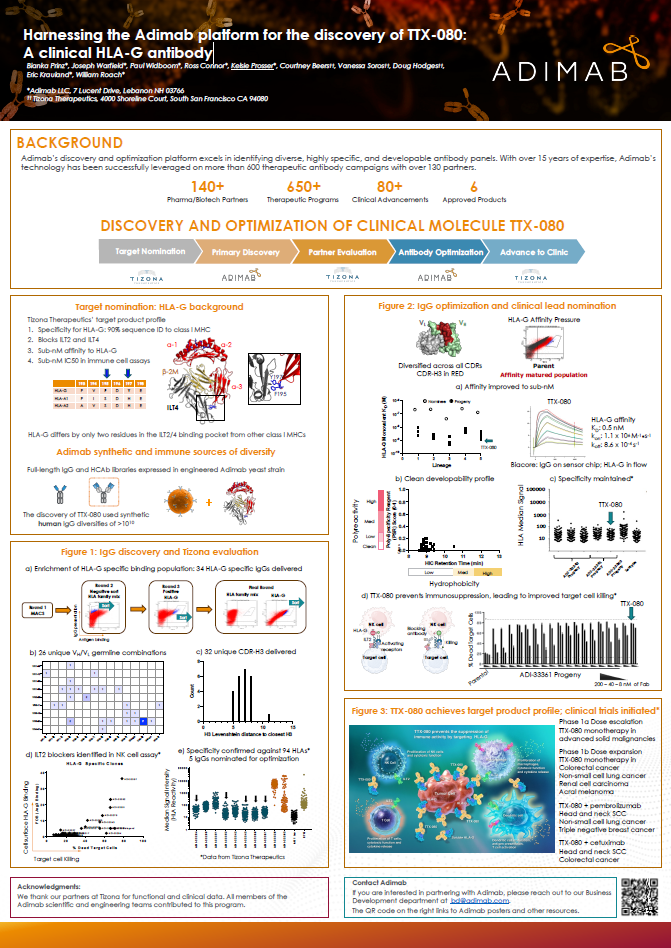
A case study describing how Adimab's platform translated HLA-G–specific leads into an antibody now in clinical trials
Transform leads into high-affinity, developable candidates through guided engineering and real-time screening.
Engineer multispecifics, including T-cell engagers, of diverse formats for function, manufacturability, and scalability.
Leverage AI-guided engineering of non-antibody proteins for tailored binding, stability, and expression.
Learn how our integrated capabilities in discovery and engineering help advance your programs to the clinic.